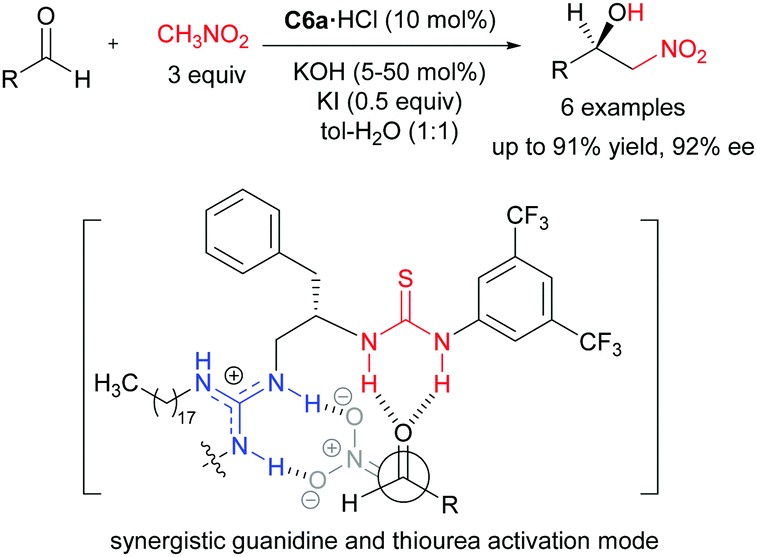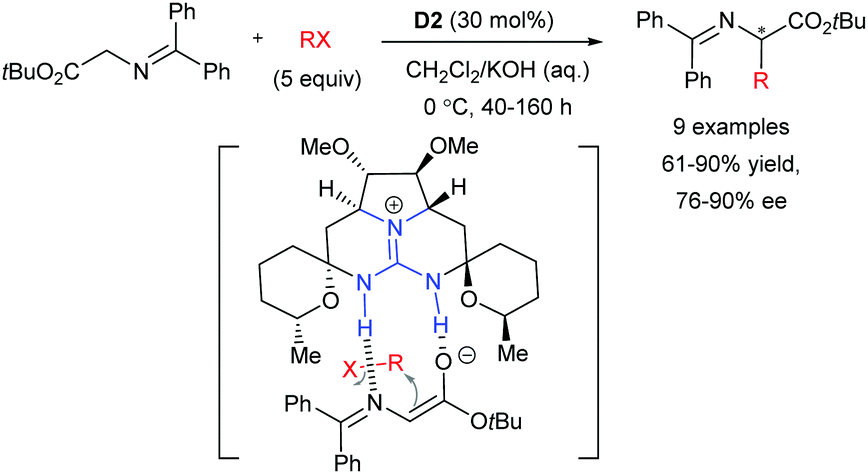
Chiral guanidines and their derivatives in asymmetric synthesis - Chemical Society Reviews (RSC Publishing)
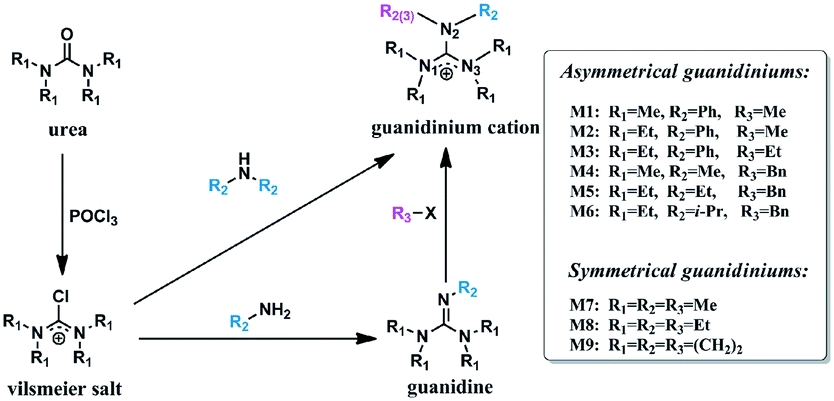
Highly stable polysulfone anion exchange membranes incorporated with bulky alkyl substituted guanidinium cations - Molecular Systems Design & Engineering (RSC Publishing)
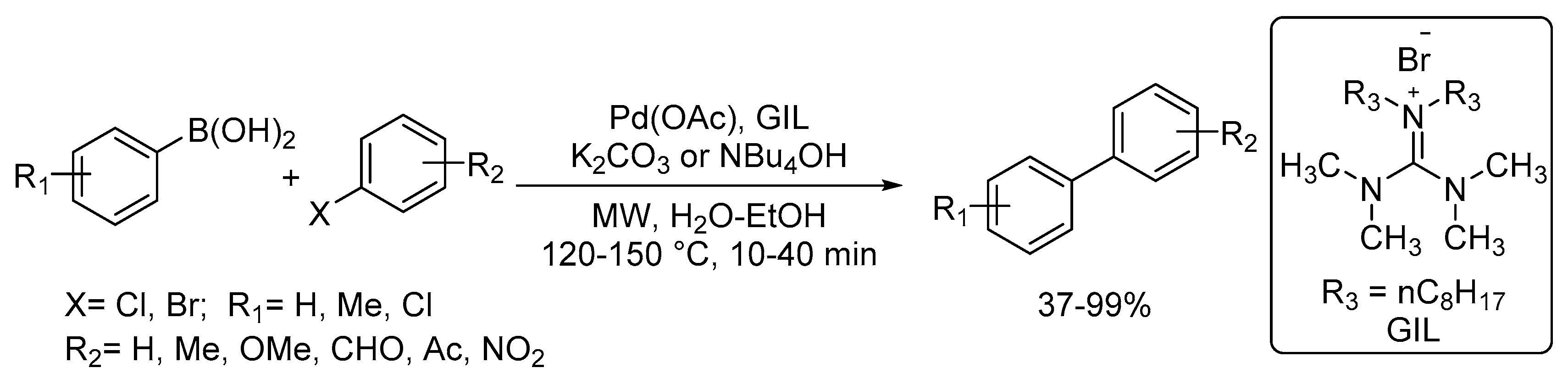
Catalysts | Free Full-Text | Microwave-Assisted Palladium-Catalyzed Cross-Coupling Reactions: Generation of Carbon–Carbon Bond | HTML

Urea derivatives from carbon dioxide and amines by guanidine catalysis: Easy access to imidazolidin-2-ones under solvent-free conditions - ScienceDirect
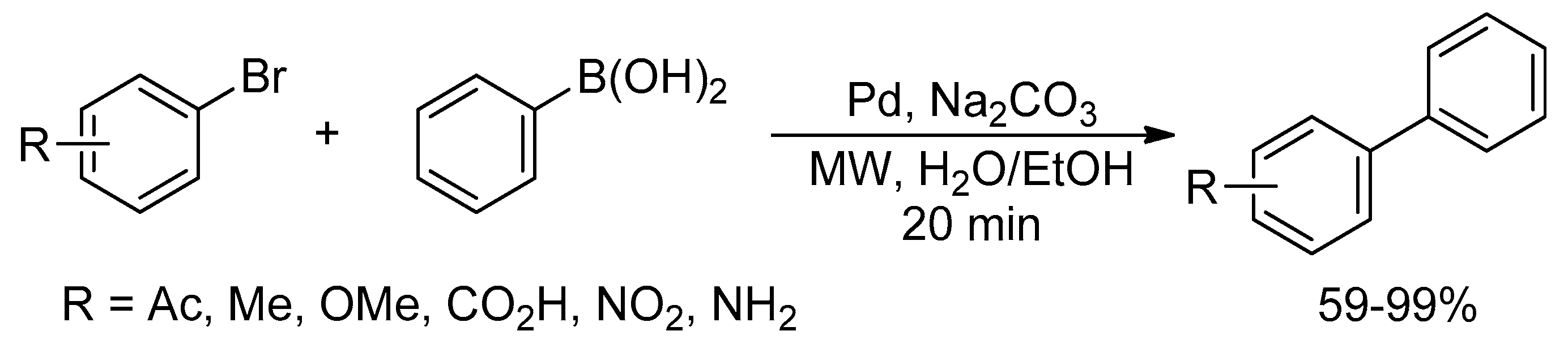
Catalysts | Free Full-Text | Microwave-Assisted Palladium-Catalyzed Cross-Coupling Reactions: Generation of Carbon–Carbon Bond | HTML

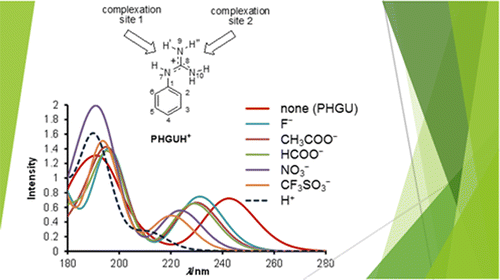
a) Molecular structures of the assembled guanidinium-functionalized... | Download Scientific Diagram
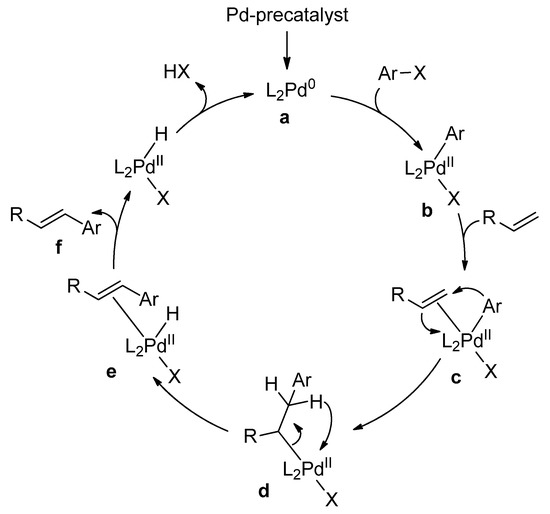
Catalysts | Free Full-Text | Microwave-Assisted Palladium-Catalyzed Cross-Coupling Reactions: Generation of Carbon–Carbon Bond | HTML
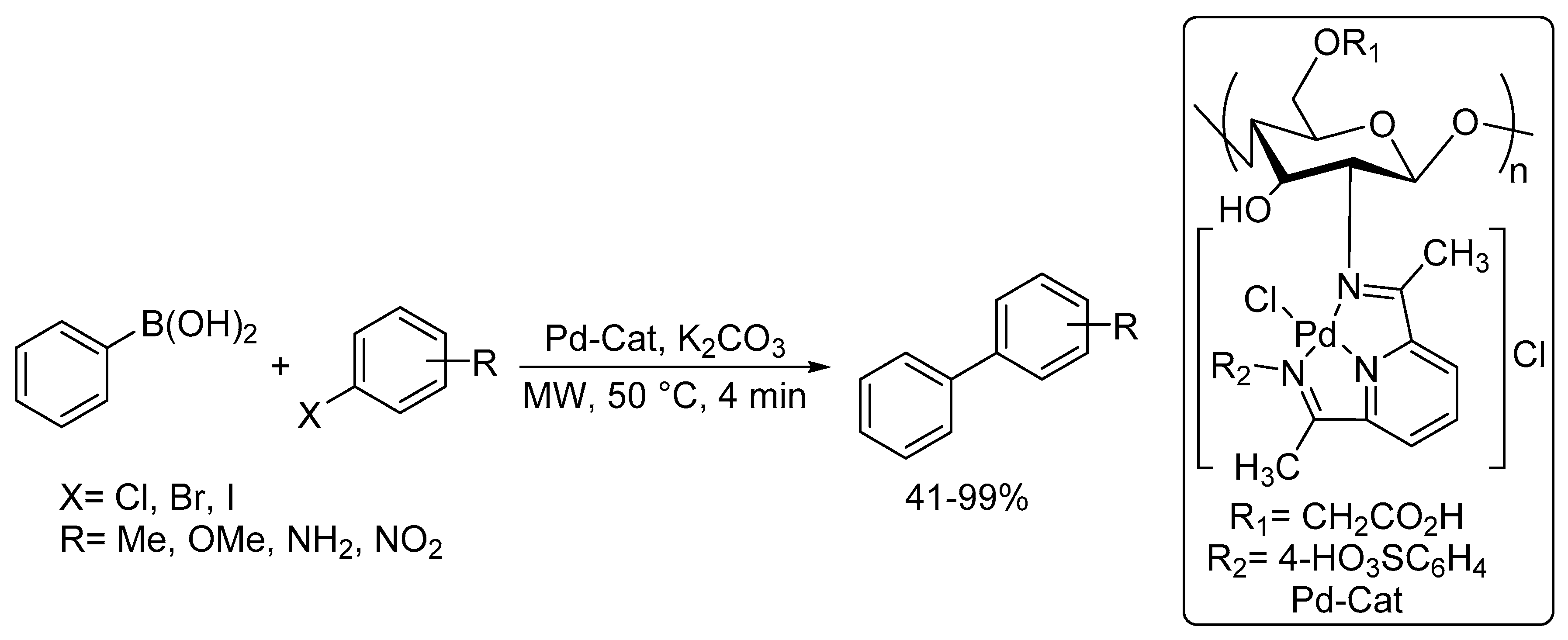
Catalysts | Free Full-Text | Microwave-Assisted Palladium-Catalyzed Cross-Coupling Reactions: Generation of Carbon–Carbon Bond | HTML
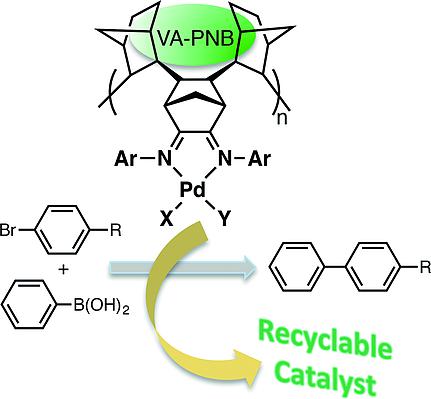
α-Diimine–Palladium Complexes Incorporated in Vinylic-Addition Polynorbornenes: Synthesis and Catalytic Activity - Eur. J. Inorg. Chem. - X-MOL

Recent Advances in Guanidine-Based Organocatalysts in Stereoselective Organic Transformation Reactions | IntechOpen
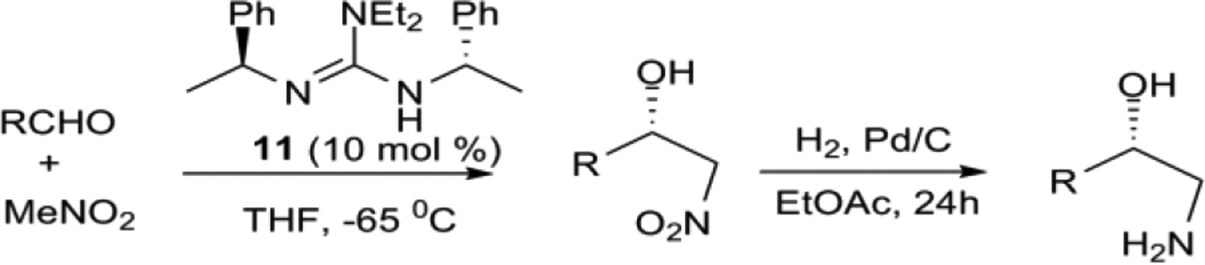
Recent Advances in Guanidine-Based Organocatalysts in Stereoselective Organic Transformation Reactions | IntechOpen
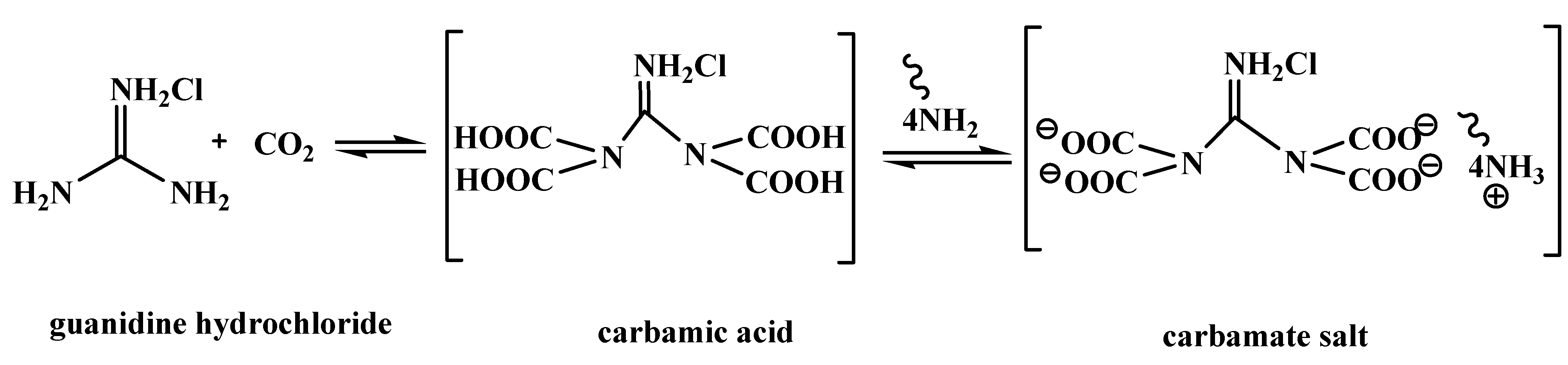
Catalysts | Free Full-Text | Guanidine Hydrochloride/ZnI2 as Heterogeneous Catalyst for Conversion of CO2 and Epoxides to Cyclic Carbonates under Mild Conditions | HTML

Recent Advances in Guanidine-Based Organocatalysts in Stereoselective Organic Transformation Reactions | IntechOpen

Guanidinium-Formamidinium Lead Iodide: A Layered Perovskite-Related Compound with Red Luminescence at Room Temperature. - J. Am. Chem. Soc. - X-MOL

Chiral guanidines and their derivatives in asymmetric synthesis - Chemical Society Reviews (RSC Publishing)
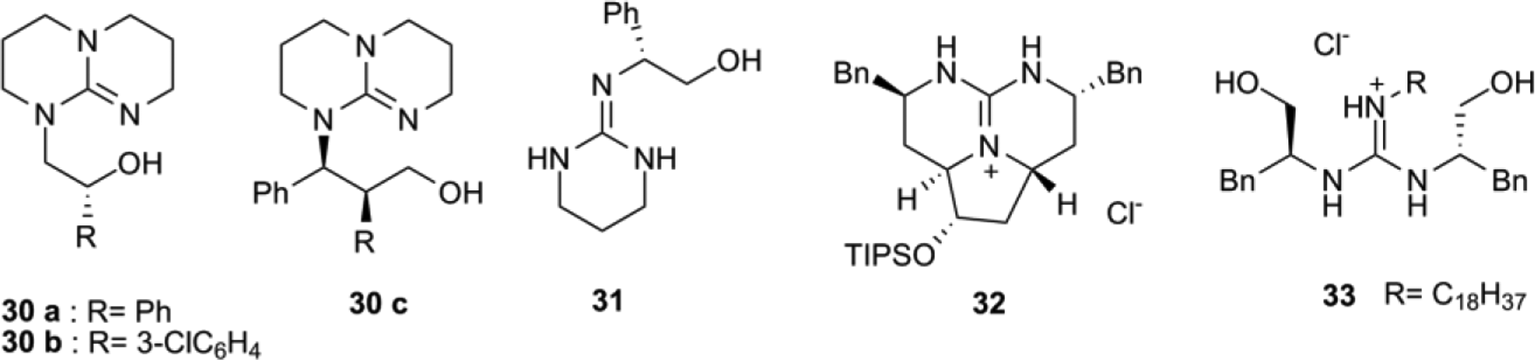
Recent Advances in Guanidine-Based Organocatalysts in Stereoselective Organic Transformation Reactions | IntechOpen
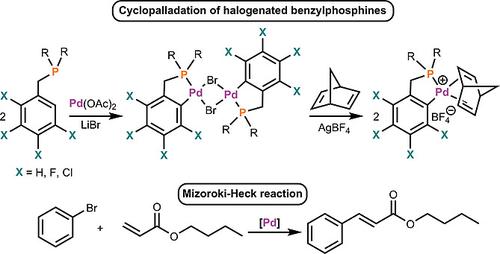
Cyclopalladated Compounds with Polyhalogenated Benzylphosphanes for the Mizoroki‐Heck Reaction - Eur. J. Inorg. Chem. - X-MOL
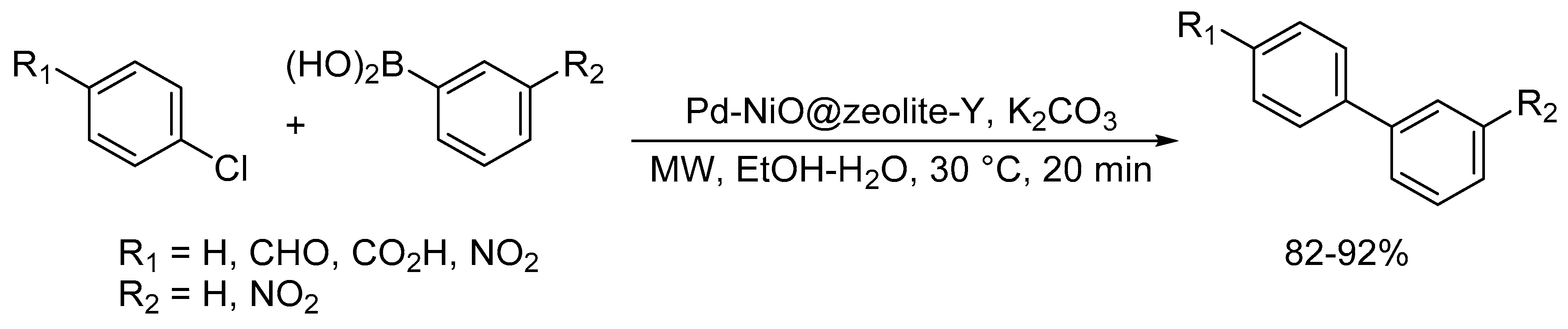
Catalysts | Free Full-Text | Microwave-Assisted Palladium-Catalyzed Cross-Coupling Reactions: Generation of Carbon–Carbon Bond | HTML
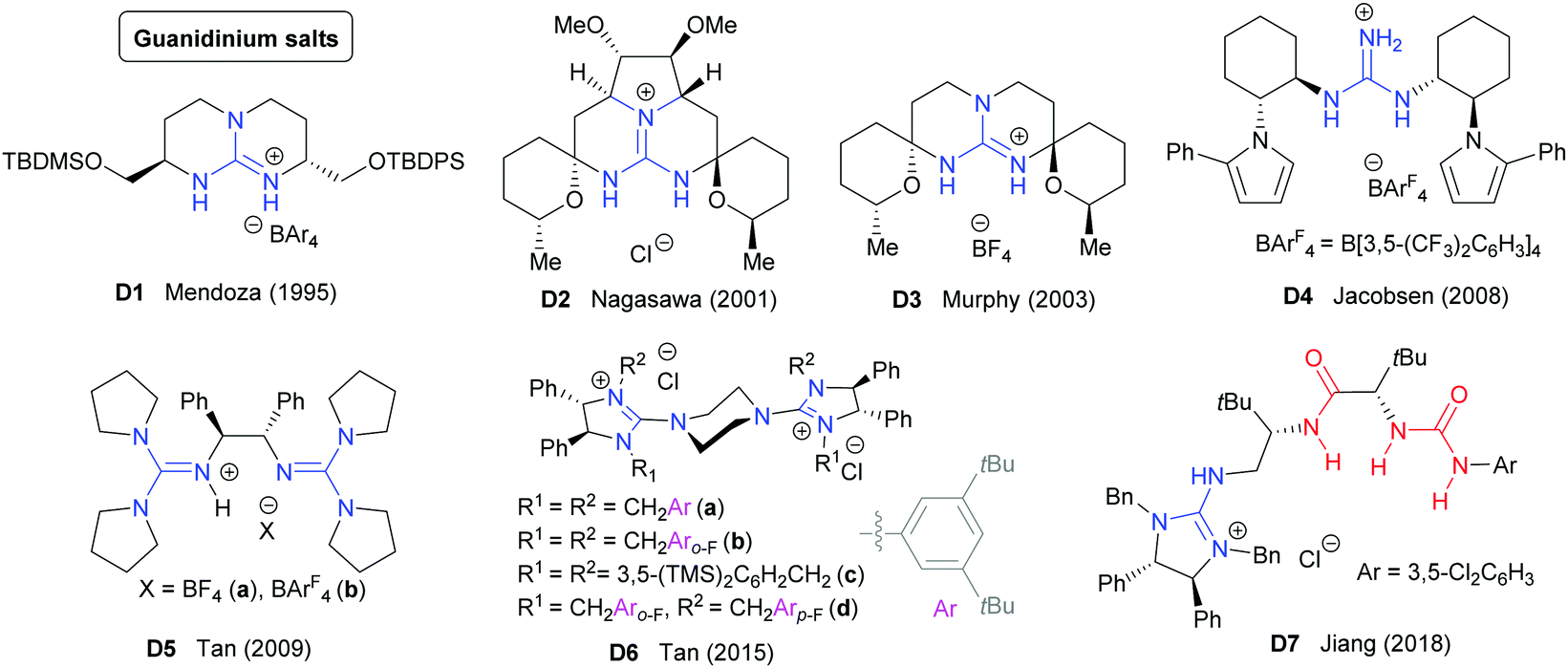
Chiral guanidines and their derivatives in asymmetric synthesis - Chemical Society Reviews (RSC Publishing)