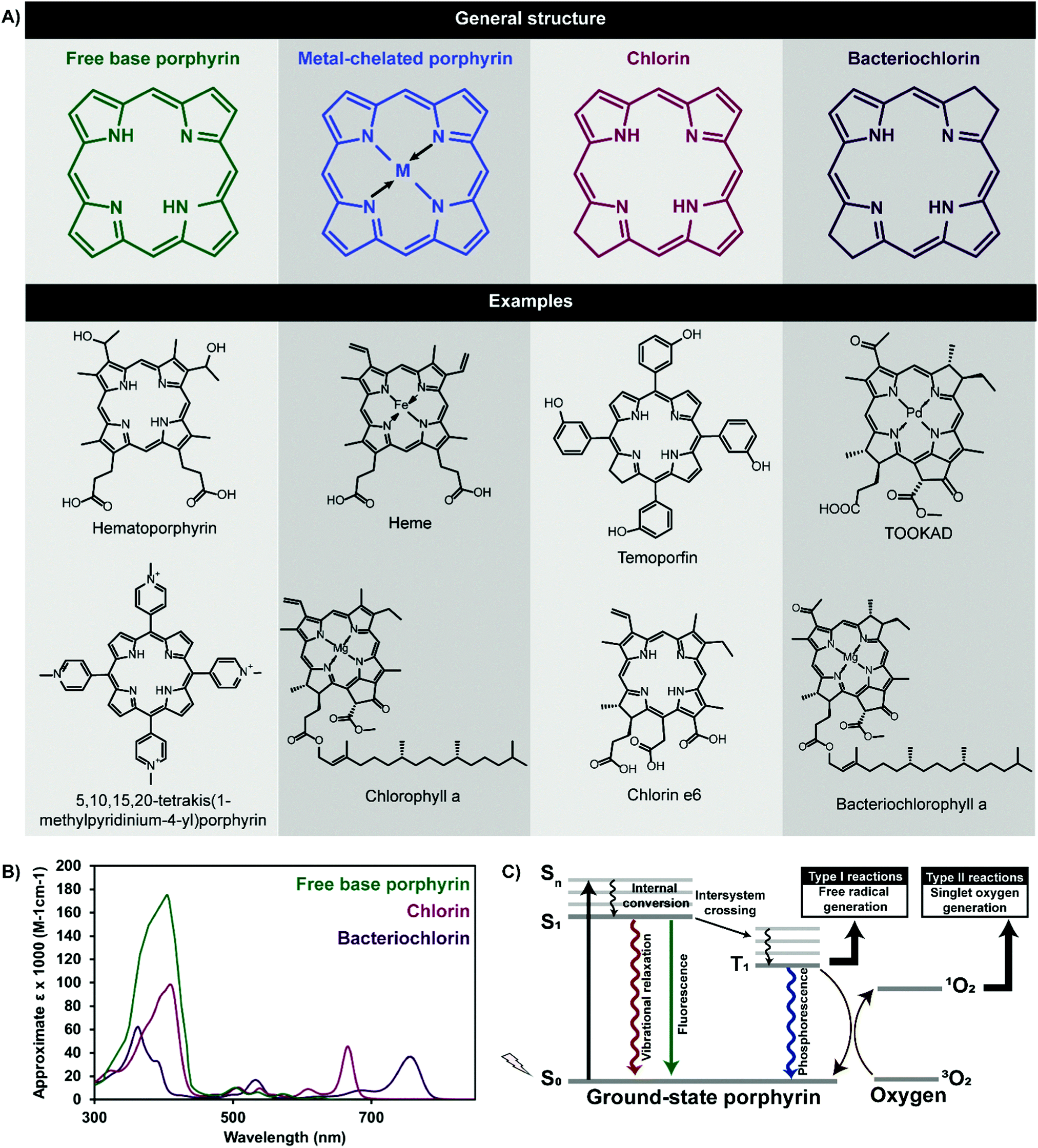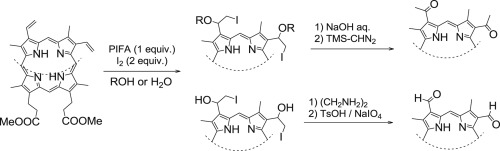
Facile iodination of the vinyl groups in protoporphyrin IX dimethyl ester and subsequent transformation of the iodinated moieties - Tetrahedron - X-MOL

US20080242857A1 - Process For Preparing Porphyrin Derivatives, Such As Protoporphyrin (IX) And Synthesis Intermediates - Google Patents

PDF) Encapsulation of palladium porphyrin photosensitizer in layered metal oxide nanoparticles for photodynamic therapy against skin melanoma

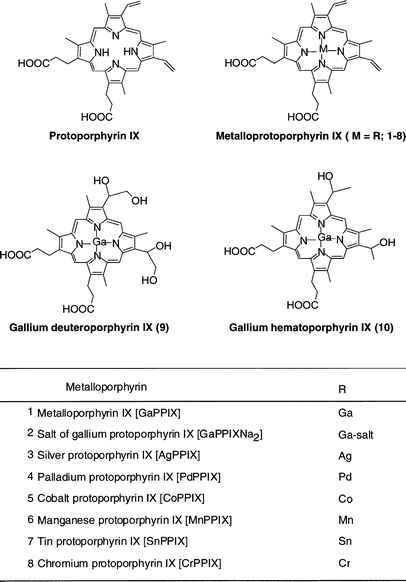
In vitro antimalarial activity of metalloporphyrins against Plasmodium falciparum | Semantic Scholar
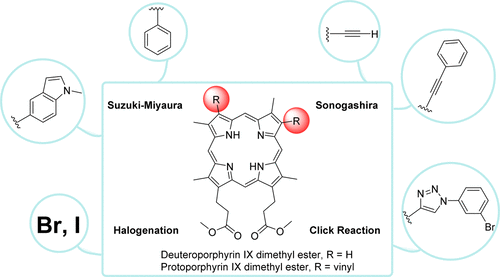
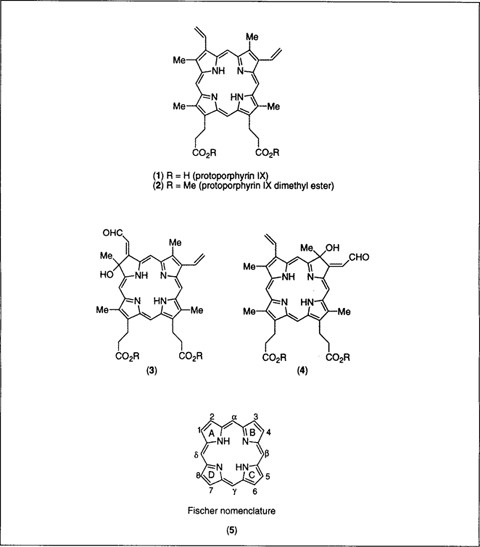
Functionalization of Deutero- and Protoporphyrin IX Dimethyl Esters via Palladium-Catalyzed Coupling Reactions - J. Org. Chem. - X-MOL

Palladium porphyrin complexes for photodynamic cancer therapy: effect of porphyrin units and metal - Photochemical & Photobiological Sciences (RSC Publishing)

PDF) Functionalization of Deutero- and Protoporphyrin IX Dimethyl Esters via Palladium-Catalyzed Coupling Reactions
![3-[18-(2-Carboxyethyl)-7,12-bis(ethenyl)-3,8,13,17-tetramethylporphyrin-21,23-diid-2-yl]propanoic acid;palladium(2+) | C34H32N4O4Pd - PubChem 3-[18-(2-Carboxyethyl)-7,12-bis(ethenyl)-3,8,13,17-tetramethylporphyrin-21,23-diid-2-yl]propanoic acid;palladium(2+) | C34H32N4O4Pd - PubChem](https://pubchem.ncbi.nlm.nih.gov/image/imgsrv.fcgi?cid=472085&t=l)
3-[18-(2-Carboxyethyl)-7,12-bis(ethenyl)-3,8,13,17-tetramethylporphyrin-21,23-diid-2-yl]propanoic acid;palladium(2+) | C34H32N4O4Pd - PubChem

5,10,15,20-Tetrakis(pentafluorophenyl)-21H,23H-porphine palladium(II) To be determined | 72076-09-6 | Sigma-Aldrich

Porphyrin Dimers and Arrays - Ryan - 2011 - European Journal of Organic Chemistry - Wiley Online Library
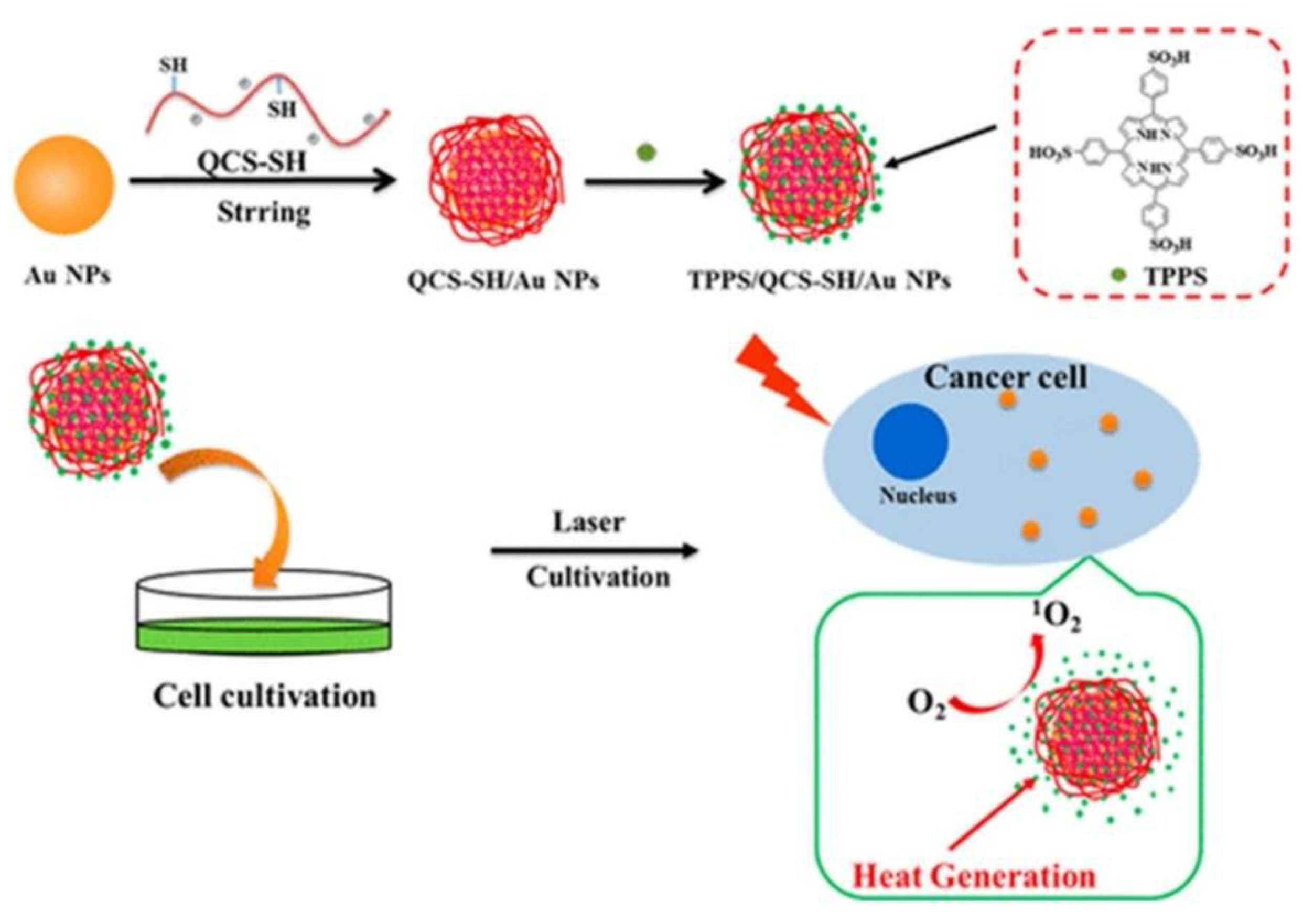
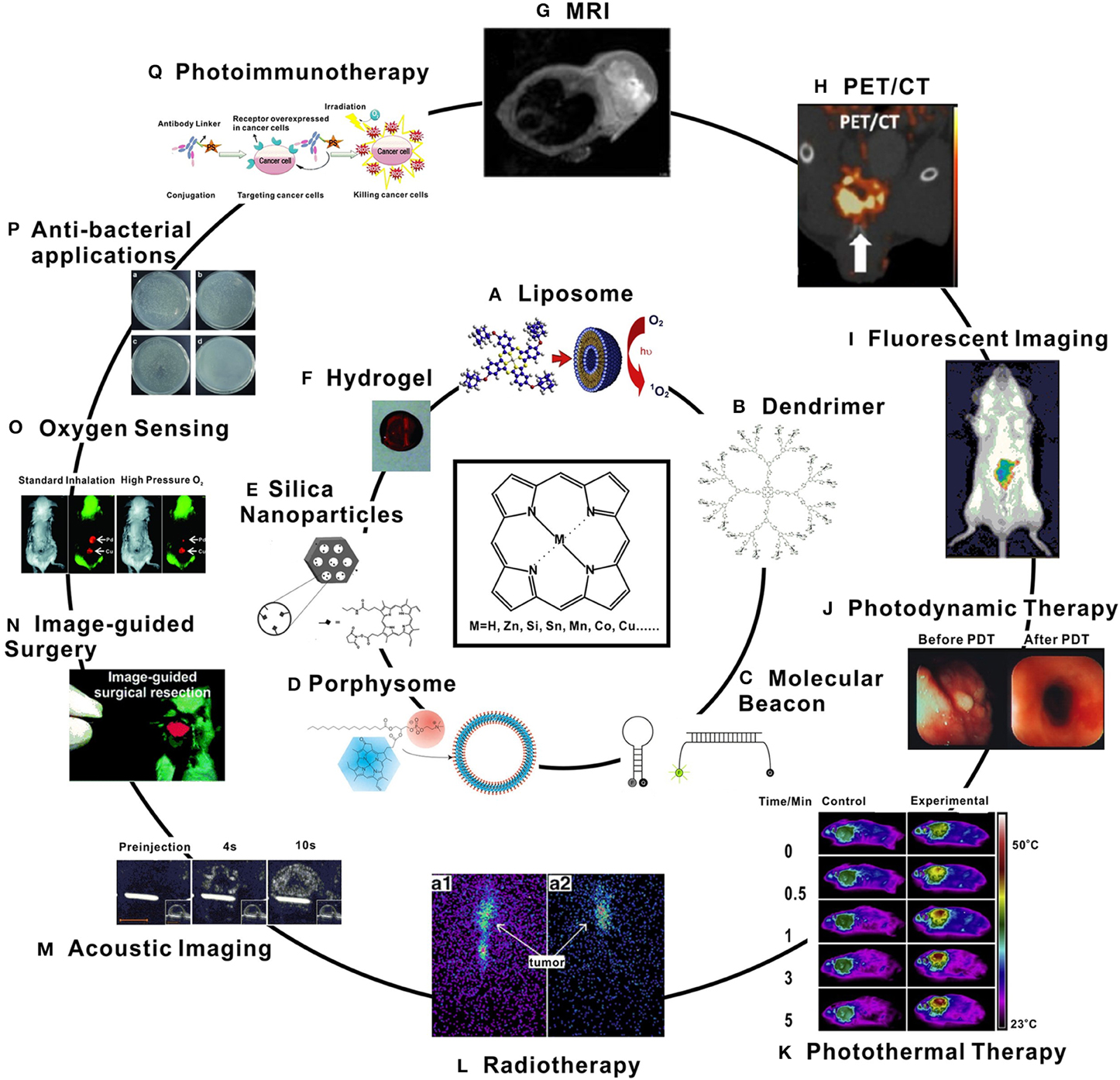
IJMS | Free Full-Text | Recent Advances in Porphyrin-Based Inorganic Nanoparticles for Cancer Treatment | HTML

PDF) Functionalization of Deutero- and Protoporphyrin IX Dimethyl Esters via Palladium-Catalyzed Coupling Reactions