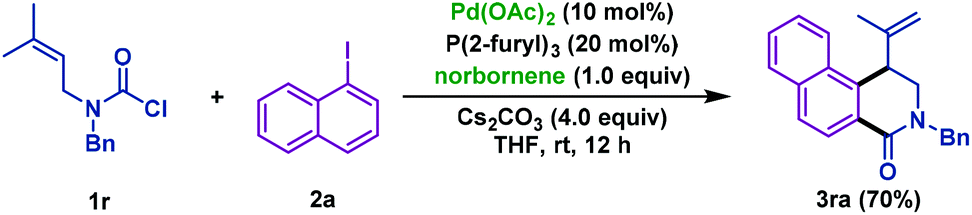
WO2013000874A1 - Method for the preparation of palladium(i) tri-tert-butylphosphine bromide dimer and process for its use in isomerization reactions - Google Patents

Palladium chemodosimeters based on change in optical properties. (a)... | Download Scientific Diagram

Convenient and General Palladium‐Catalyzed Carbonylative Sonogashira Coupling of Aryl Amines - Wu - 2011 - Angewandte Chemie International Edition - Wiley Online Library

PDF) Sequential and cascade palladium catalysed cyclisation-anion capture-olefin metathesis | Mark York - Academia.edu
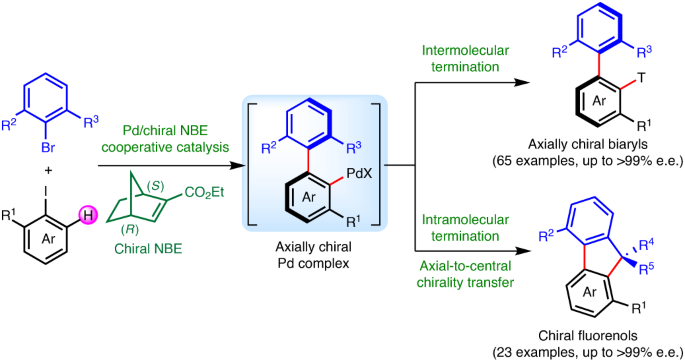
Construction of axial chirality via palladium/chiral norbornene cooperative catalysis | Nature Catalysis
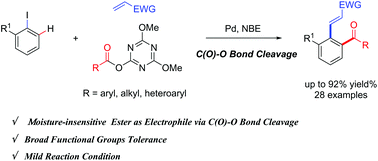
A practical ortho-acylation of aryl iodides enabled by moisture-insensitive activated esters via palladium/norbornene catalysis - Organic Chemistry Frontiers (RSC Publishing)

PDF) Selective palladium-mediated synthesis of racemic 4,5-disubstituted 5H- furan-2-ones from 3-ynoic acids and organic halides | Fabio Bellina - Academia.edu

WO2014175841A1 - Palladium complexes of curcumin and its analogues and methods of preparation of the same - Google Patents
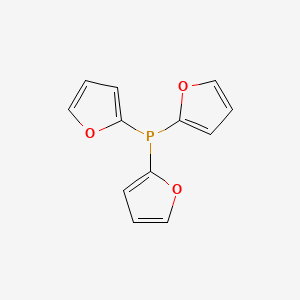
![5518-52-5・Tri(2-furyl)phosphine・202-18631・208-18633[Detail Information] | [Synthesis & Materials] |Laboratory Chemicals-FUJIFILM Wako Chemicals U.S.A. Corporation 5518-52-5・Tri(2-furyl)phosphine・202-18631・208-18633[Detail Information] | [Synthesis & Materials] |Laboratory Chemicals-FUJIFILM Wako Chemicals U.S.A. Corporation](https://labchem-wako.fujifilm.com/sc/01/5518-52-5.png)
5518-52-5・Tri(2-furyl)phosphine・202-18631・208-18633[Detail Information] | [Synthesis & Materials] |Laboratory Chemicals-FUJIFILM Wako Chemicals U.S.A. Corporation

Recent advances in the use of tri(2-furyl)germane, triphenylgermane and their derivatives in organic synthesis - ScienceDirect

Recent advances in the use of tri(2-furyl)germane, triphenylgermane and their derivatives in organic synthesis - ScienceDirect